 New CDC guidelines say that people who have been vaccinated against COVID-19 can go without masks in indoor settings with others who have also gotten their shots. But vaccinated individuals can also visit unvaccinated people in some scenarios, too, meaning vaccine-protected grandparents can hug their unvaccinated grandkids. JOSE LUIS PELAEZ INC/GETTY IMAGES PLUS Share this:By Erin Garcia de Jesus and Tina Hesman Saey MARCH 8, 2021 AT 2:49 PM As the pace of coronavirus vaccinations picks up, normal life is looking tantalizingly in reach. Nearly a year after the World Health Organization declared the coronavirus outbreak a pandemic (SN: 3/11/20), and social distancing efforts sent billions of people into isolation, the U.S. Centers for Disease Control and Prevention is outlining some of the social perks to getting vaccinated. Vaccinated friends can have dinner together in their homes without wearing masks or physical distancing, CDC director Rochelle Walensky said March 8 during a White House coronavirus briefing. “You can visit your grandparents if you have been vaccinated and they have been, too.” But vaccinated people still need to take precautions around the unvaccinated, especially those at high risk of getting severely ill from COVID-19, and group settings are still considered risky. Sign up for e-mail updates on the latest coronavirus news and researchThese guidelines are a first step toward letting people know what aspects of normal life can resume, Walensky said, but could change as new data become available. For instance, the guidelines could become more permissive as vaccination becomes more widespread and cases continue to fall. But if new coronavirus variants that can reinfect even people who have previously had COVID-19 take hold, restrictions might be reimposed. The news comes as about 2 million coronavirus vaccine shots are being delivered into people’s arms in the United States each day. As of March 8, 59 million adults in the United States have gotten at least one dose of a COVID-19 vaccine. Of those, 31 million people — 9.2 percent of the U.S. population — are fully vaccinated. That means it’s been at least two weeks since they’ve gotten both doses of the Pfizer or Moderna vaccines or the single shot of the Johnson & Johnson vaccine. Those numbers are “putting us on a strong path to eventually ending this pandemic,” Walensky said. While results from clinical trials have indicated that all of the vaccines protect against severe illness and death, there is still a small risk that vaccinated people might get mild or asymptomatic infections, which they might pass on to unvaccinated people, she said. Some data indicate that the Pfizer vaccine lowers the risk of infection and may reduce the risk of transmitting the virus to others (SN: 2/26/21; SN: 2/12/21), but definitive studies have yet to be completed. Other vaccines may also lower the risk of catching and passing along the virus, but there is less known about those shots. See all our coverage of the coronavirus outbreakStill, “as more Americans get vaccinated, a growing body of evidence tells us that there are some activities that fully vaccinated people can resume at low risk to themselves,” Walensky said. Fully vaccinated people can start visiting also vaccinated friends and family two weeks after their last jab. For these vaccinated groups, indoor gatherings sans masks or social distancing are OK, according to the new guidelines. Plus, those individuals don’t need to quarantine if exposed to the coronavirus, as long as they don’t have any COVID-19 symptoms. It gets trickier when unvaccinated people are thrown into the mix, particularly those at high risk of severe illness or death — including those 65 and older and people with health conditions such as heart disease, diabetes or cancer that make them more vulnerable to complications, Walensky said. Socializing with unvaccinated high-risk individuals and their family members still isn’t recommended. But meet-ups among vaccinated people and people from one household who are unvaccinated but at low risk for becoming severely ill if infected with the coronavirus are fine. Sign Up For the Latest from Science NewsHeadlines and summaries of the latest Science News articles, delivered to your inbox E-mail* GOVaccinated people should still avoid larger gatherings, including mask-less get-togethers with more than one unvaccinated household. Vaccinated individuals also shouldn’t meet with someone who is unvaccinated and at high risk for severe COVID-19, or with a high-risk person’s family members. In these scenarios, everyone should still wear a mask and keep their distance. And while out in public, vaccinated people should still continue to wear well-fitting face masks and keep their distance from others (SN: 2/12/21). That helps further reduce the coronavirus’s spread and protect others who haven’t gotten shots yet. The CDC also still recommends putting off travel and staying home for now. Questions or comments on this article? E-mail us at [email protected]
0 Comments
 Antibodies made in the lab show some promise for treating COVID-19 New results hint that the proteins can reduce hospitalizations or need for ventilationPreliminary results released by two companies suggest that lab-made monoclonal antibodies (Y-shaped proteins in this 3-D illustration) may help treat COVID-19 patients. By Erin Garcia de Jesus SEPTEMBER 22, 2020 AT 3:40 PM Amid the rush to test and develop potential treatments for COVID-19, lab-made antibodies are showing hints of success. In news releases, two companies announced preliminary results, though shared only limited data, that suggest the experimental drugs may help patients both early and late in infection. One clinical trial of monoclonal antibodies — human-made versions of immune system defenders produced by the body — suggests that the drugs can help keep people hospitalized with COVID-19 from needing a ventilator or from dying. And a second trial appears to show that the drugs can bring down levels of the coronavirus in recently infected people, and help reduce the chances that a person would need hospitalization. Antibodies are part of the body’s natural defense against infectious pathogens. The proteins typically attach to parts of bacteria or viruses to fight off infection. In the lab, scientists can engineer versions of antibodies to recognize specific targets in order to hinder the virus’ replication or prevent the body’s immune system from overreacting to the virus (SN: 2/21/20). Sign up for e-mail updates on the latest coronavirus news and researchA monoclonal antibody drug called tocilizumab is one of the latter types; it blocks a part of the immune response that can cause inflammation, a protein known as IL-6. By curbing inflammation, the drug could help people whose immune systems have become overactive through a process called a cytokine storm, which can cause severe COVID-19 symptoms (SN: 8/6/20). In a Phase III clinical trial of 389 people hospitalized with COVID-19, those who received tocilizumab were 44 percent less likely to need a ventilator or die compared with people who got a placebo, San Francisco–based biotechnology company Genentech announced September 17 in a news release. Of those who received the drug, 12.2 percent of people needed a ventilator or died, compared with 19.3 percent of patients who received a placebo. Still, when the researchers looked at death alone, the drug did not result in a statistically significant difference in mortality between the groups. Saying that it was still analyzing the data, the company did not provide such specifics as how many people died in each group. “A 44-percent decrease is definitely very intriguing,” says Abhijit Duggal, a critical care specialist at the Cleveland Clinic who has treated people with COVID-19. But because the results have been publicized in a news release, without key patient information, “I don’t know what to really make of that,” Duggal says. Only as more data come in will experts be able to conclusively say whether the drug might help people, he says. The announced results have not yet been vetted by outside experts or published in a peer-reviewed journal. Unlike many other clinical trials of potential COVID-19 drugs and treatments, the Genentech trial focused on groups of people that have been disproportionately impacted by the virus (SN: 4/10/20). Around 85 percent of people in the study are Black, Hispanic and Native American. People in these groups are more likely than white people to be infected or die from COVID-19, studies have shown. In part that’s due to high rates of underlying conditions like high blood pressure and jobs with a higher risk of exposure to the virus. “It’s really important that [the researchers] are including a diverse population,” says Rajesh Gandhi, an infectious disease physician at Massachusetts General Hospital and Harvard Medical School in Boston. “That is critical as we do these trials.” GOIn a previous Genentech-related trial that included 452 people with severe COVID-19, tocilizumab did not help improve symptoms or prevent death, researchers reported in a preliminary study posted September 12 at medRxiv.org. Other trials of the drug have reported improved outcomes in people with moderate or severe COVID-19 symptoms. Importantly, the new trial focused on hospitalized people before they required a ventilator, says Jamie Freedman, Genentech’s head of U.S. medical affairs. So differences among trials could be a timing issue. “If you give it too early, before cytokines are elevated, would there be a benefit there? When patients are already in the ICU, is it too late?… Or is there some sweet spot in the middle?” Freedman says. “Those are analyses that really need to continue.” Scientists working on another monoclonal antibody, which targets the coronavirus’ spike protein, also recently reported promising results (SN: 2/21/20). Called LY-CoV555, the drug can reduce the amount of virus in the bodies in newly infected people and help prevent COVID-19 hospitalizations, Indianapolis-based pharmaceutical company Eli Lilly announced September 16 in a news release. People in this ongoing Phase II clinical trial to determine efficacy receive either a low, medium or high dose of the antibody or a placebo. So far, those who get a medium dose of LY-CoV555, which is based on an antibody from one of the first COVID-19 patients in the United States, appear to clear the virus faster than those on the placebo, according to the release. Fewer treated patients still had high viral loads later on in the study. Most people, including those on a placebo, cleared the virus from their bodies by day 11. Like Genentech, Eli Lilly released only limited data. The announced results have not yet been vetted by outside experts or published in a peer-reviewed journal. See all our coverage of the coronavirus outbreak“It’s really intriguing and tantalizing information,” Gandhi says. But without the full details of the study, like patient age or whether any people had underlying conditions, it’s difficult to know how solid the findings are, he says. It’s surprising that people on the medium dose had a benefit from the drug but those on the higher dose didn’t, but that could be because the results are preliminary and could change as people are added to the trial, says Nina Luning Prak, an immunologist at the University of Pennsylvania. “But in principle, it looks hopeful,” she says. What’s more, of 302 people treated with any amount of LY-CoV555, five, or 1.7 percent, landed in the hospital, while nine people, or 6 percent, in a control group of 150 patients who received a placebo, were hospitalized. It’s unclear based on the results included in the news release, however, whether the difference between the two groups is meaningful. But “if it’s borne out, we’ll see — hopefully soon — that this is important because it shows that an antibody is having an antiviral effect,” Gandhi says. There are many other monoclonal antibody trials ongoing around the world, many of which feature drugs that bind to a variety of both virus and host proteins. Experts are carefully watching for results, keen to know for sure whether such treatments can help patients. Still, compared with where treatments were in March and April, “we’ve made progress,” Gandhi says. “I think that progress is going to just accelerate.”  This super-strong adhesive can be undone by adding water ALISON PEARCE STEVENS OCT 15, 2019 — 6:45 AM EST When not on the move, snails use mucus to stick to plants and other objects. Makers like to combine unusual items to create new things. To do so, they need super-strong adhesives — tapes and glues — to hold it all together. But sometimes they want to be able to take the items apart again. That’s been a problem, because reversible adhesives usually are not very strong. Sticky stuff can be super-strong and permanent — like superglue. Or it can be less sticky but easily removed — think of a sticky note. Now, though, researchers have created an adhesive that’s both reusable and super-strong. Shu Yang works at the University of Pennsylvania in Philadelphia. Her team described their new superglue July 9 in the Proceedings of the National Academy of Sciences. As a materials scientist, Yang uses physics, chemistry and engineering to create new types of items. In her work, Yang often finds inspiration for new materials based on structures that exist in nature. For years, she has been working to create an adhesive that not only holds well but also can be undone and reused. Earlier work mimicked the tiny hairs on gecko feet. Although the material came unstuck easily, it didn’t have a strong grip. So the scientists in Yang’s lab kept searching for something better. Explainer: What are polymers?One day, a student in her lab was playing with a substance known as a hydrogel. A polymer, it’s made up of repeating chains of smaller chemicals. This particular gel turns soft when wet — in fact, it’s what makes contact lenses so flexible. Yang’s lab had been using it to make various structures for about 10 years. The student, Gaoxiang Wu, made patterns with it on a glass slide and then left it there. When Wu returned, the hydrogel had hardened and was seriously stuck. He pulled, pried and scraped, but nothing separated the gel from the glass slide. Then he added water — and it came right off. That finding made Yang and her team curious. Why had this dried gel been so hard to remove? They also wondered whether anything in nature might also work that way. And before long they discovered that snails make a similarly sticky goo. During the heat of the day, snails are at risk of drying out. To prevent this, a snail finds a good spot near the ground with plenty of moisture. There, it pumps lots of mucus through the opening on its shell. The mucus oozes over the ground, filling in any gaps. As it dries, the mucus hardens. This creates a structure that is both protective and adhesive. Called an epiphragm (EP-ih-fram), it seals the moist snail inside its shell, protecting it from predators that would readily munch on it if they could get to the meat inside. When temperatures fall in the evening and humidity rises, the mucus loosens. Now free to move, the snail continues on its way. Yang and her team reached out to Anand Jagota. He is a bioengineer at Lehigh University in Bethlehem, Penn. Jagota specializes in soft materials and their adhesive properties. Together, the researchers studied the hydrogel and found that it worked the same way as a snail’s slime. When the gel was wet, it oozed into every little nook and cranny, just like snail mucus. When it dried, the material turned hard and glassy. Now it was almost impossible to pull off. The key, Yang says, is that the gel is very soft when wet. It’s a lot like those squishy splat balls that stick to the wall, she says. Almost every surface has tiny imperfections, she observes. So with just a small bit of pressure, the gel can squeeze into all of those the tiny pores and spaces on the surface it’s sticking to. For most adhesives, that surface roughness is a problem. It reduces the amount of contact between the adhesive and surface, she notes. That makes the adhesive less sticky. But the hydrogel fills in those itty-bitty gaps, forming a tight connection. What’s more, the gel doesn’t shrink much as it dries. So it doesn’t pull away from those rough patches. Instead, it holds its shape as it turns glassy. In fact, that’s key to why it sticks so well — it’s locked on to the tiny cavities. “We couldn’t separate the gel from the substrate,” Yang says. No matter what tool they tried. Strong adhesives are important for making things — building cars or airplanes, for example. So the team wanted to figure out just how strong their new adhesive was. Scientists Say: KevlarTo find out, they made the gel on one side of a sheet of silicon. They cut the gel-coated silicon into small chips no bigger than a postage stamp. Then they superglued a piece onto each side of a strip of heavy-duty metal. They glued other squares of bare silicon to one end of each of two Kevlar ribbons. (Kevlar is a tough material used to make bulletproof vests. It wouldn’t fall apart during testing.) Finally, the team added water to the hydrogel and pressed the ribbons’ bare silicon wafers onto either side of the gel-coated metal strip. When it dried, the Kevlar ribbons were firmly attached to the metal. Yang’s team used the hydrogel superglue to attach Kevlar ribbons (called tendons) to a metal plate. They used this setup to test how strong the glue is. Si-wafer refers to a chip made of silicon. H. Cho et al./Proceedings of the National Academy of Sciences 2019The team used the hydrogel to attach the metal strip to the front of a filing cabinet. They then hung a 4-kilogram (8.8-pound) weight from the Kevlar ribbon. It held firmly. Only when the team added water did the hydrogel soften, letting the weight fall to the ground. The hydrogel was even stronger than the superglue. Sometimes when the weight was added, the superglue used in the setup broke free. But the hydrogel always held fast. The new super-adhesive is so strong that two postage-stamp-sized pieces can hold up an adult. H. Cho et al/Proceedings of the National Academy of Sciences 2019In a final test, the team built a large metal frame and stuck the gel-coated strip with the Kevlar ribbons to the top. They attached the Kevlar to a harness worn by a student in the lab. He carefully lifted his feet off the ground — and waited. The strip held. Just two postage-stamp-sized pieces of gel held up an 87-kilogram (192-pound) man! The new adhesive “beautifully imitat[es] the reversible bonding mechanism used in the snail’s epiphragm,” says Diana Kay Hohl. She is a materials scientist at the University of Fribourg in Switzerland. Water is a good choice to make the new adhesive non-toxic, she says. But water is slow to dry. The hydrogel takes longer to dry than many other strong glues, Hohl notes. And water won’t work in many types of manufacturing, she points out. “It will be interesting to see if, in next-generation materials, the adhesion can be tuned by triggers other than water,” she says. Heat or light, for example, might make the super-adhesive more suitable for manufacturing.  FROM NPR: Updated at 3:30 p.m. ET About 35 years after the first American woman walked in space, NASA set another milestone on Friday, as astronauts Christina Koch and Jessica Meir conducted the first all-female spacewalk. Koch, an engineer, and Meir, a biologist, suited up and entered the "crew lock" that serves as the International Space Station's doorway into space. They switched on the battery power in their suits at 7:38 a.m. ET, signaling the official start of the spacewalk. The outer hatch began opening as controllers on the ground finished ensuring the astronauts' suits were ready, including visors that are required because of the sunlight hitting the station. Then, Koch received the word. "Christina, you may egress the airlock," said NASA's Stephanie Wilson, the spacewalk's coordinator at Mission Control Center in Houston. "Copy," Koch answered. Astronauts Christina Koch and Jessica Meir conducted some much-needed work on the exterior of the International Space Station on Friday. Along the way, they also enjoyed amazing views of Earth. NASAIn what the space agency billed as "HERstory," the four men aboard the International Space Station remained inside the orbital outpost, as Koch and Meir stepped outside to replace a broken battery charger. The pair's spacewalk went nearly two hours longer than their planned 5.5-hour excursion. After finishing replacing the battery component in about three hours, they moved on to other tasks, such as installing a stanchion to support a new research platform that will be used on the outer surface of the station next year. All the while, they worked to keep their safety tethers from becoming twisted as they moved around. More than six hours after the astronauts exited the space station, they received word from Mission Control that their work on the battery was showing good results, triggering relief as they moved on to more work. In all, their spacewalk lasted 7 hours and 17 minutes. At the start of their outing, the astronauts took a moment to get used to moving around in the vacuum of space — dubbed a "translation adaptation." They then set about preparing their tools for replacing a failed power controller in the system that collects and distributes solar power to the space station's systems. The charger failed after Koch's last extravehicular activity, or EVA, as spacewalks are known in NASA speak, when she and fellow astronaut Andrew Morgan installed new batteries on the outside of the station last week. The historic all-female spacewalk has been months in the making. It was originally supposed to happen in March, with astronaut Anne McClain joining Koch. But the station was short one functioning, medium-sized spacewalking suit, the right size for both women. Koch went instead with colleague Nick Hague. Be sure to watch live: https://nasa.gov/live "As much as it's worth celebrating the first spacewalk with an all-female team, I think many of us are looking forward to it just being normal," veteran astronaut Tracy Caldwell Dyson said on NASA's live coverage of the spacewalk. "I think if it signifies anything," she added, "it is to honor the women who came before us, who were skilled and qualified — and didn't get the same opportunities we get today because it is so normal." Koch, an electrical engineer, had done previous spacewalks, including her first with Hague in March. She's seven months into a planned 11-month stay on the station. Friday's EVA is a first for Meir, who is also on her first spaceflight. NASABy forming an all-female team, Koch and Meir's EVA is a first for any nation in space. "With today's first all-woman spacewalk on the International Space Station led by NASA astronauts Christina Koch and Jessica Meir, America once again sets a precedent for women to lead in human space exploration," NASA Administrator Jim Bridenstine said of the historic event. Bridenstine was part of a conference call from the White House, along with President Trump, Vice President Pence and the president's daughter and adviser, Ivanka Trump. "What you do is incredible. It's so — you're very brave people," President Trump said. "I don't think I want to do it, I must tell you that. But you are amazing people." "This is truly historic," Trump added. "Thank you — we don't want to take too much credit," Meir replied, noting the other women who have performed spacewalks in the past. "For us, this is really just us doing our jobs." SPACEFor NASA's New Suits, 'Mobility' Is The WatchwordSally Ride became America's first woman in space (aboard Space Shuttle Challenger) in 1983, 20 years after Russian cosmonaut Valentina Tereshkova was launched into orbit on Vostok 6. A year after Ride's first flight, another Russian woman, Svetlana Savitskaya, became the first woman to walk in space, followed a few months later, on Oct. 11, 1984, by America's first female spacewalker, Kathryn Sullivan. Meir is the 15th female spacewalker; by comparison, 213 men have walked in space, according to The Associated Press. The first person to walk in space was Soviet cosmonaut Alexei Leonov, who died last week. After he set the mark in March 1965, he was followed less than three months later by American Ed White. Science News-Monday Sept. 23 2019 Mucus Prevents handsanitizer from quickly killing the flu9/23/2019  Flu viruses can hold out for minutes against ethanol when encased in wet mucusAlcohol-based hand sanitizers used in medical settings might not protect against spreading the flu when mucus is involved. By Tina Hesman Saey SEPTEMBER 18, 2019 AT 1:00 PM Sticky mucus may thwart alcohol-based hand sanitizers’ ability to fight the flu. Flu viruses encased in mucus drops from infected people’s spit can withstand the alcohol in hand sanitizers for more than two minutes, researchers report September 18 in mSphere. Researchers dotted volunteers’ fingers with either mucus or saline solution containing the flu virus, then measured how long it took to inactivate the virus in both wet and dry samples. A five-microliter drop — about the size of a pinhead — of mucus-coated flu virus took more than half an hour to dry, almost twice as long as saline. Drying time was important because previous studies tested sanitizers’ killing ability on dry viruses, and didn’t account for mucus’s moistening power. A hand sanitizer containing 31 percent alcohol inactivated flu viruses in saline solution within 20 seconds. And in already dried mucus, that process took just under eight seconds. But moist mucus shielded flu viruses from alcohol, keeping the viruses viable for up to 2 minutes and 39 seconds, Ryohei Hirose, an infectious disease researcher at the Kyoto Prefectural University of Medicine in Japan, and his colleagues found. That may be long enough for health care workers to unwittingly transfer virus-infected mucus from one person to another. The team didn’t test whether rubbing the sanitizer over the skin causes the alcohol to penetrate the mucus and kill viruses faster, Hirose says. Rubbing might help, he acknowledged. But there’s already an easy way to kill flu viruses: Washing hands with plain water or with soap killed viruses within 30 seconds, even when the mucus was still wet. Questions or comments on this article? E-mail us at [email protected] CITATIONSR. Hirose et al. Situations leading to reduced effectiveness of current hand hygiene against infectious mucus from influenza virus-infected patients. mSphere. Published online September 18, 2019. doi: 10.1128/mSphere.00474-19. 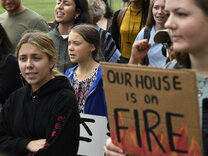 A Rising Generation Asserts Itself On Climate Change September 20, 20195:05 AM ET JEFF BRADY Swedish climate activist Greta Thunberg (center) marches with other young climate activists last week outside the White House in Washington, D.C. Susan Walsh/APSpurred by what they see as a sluggish, ineffectual response to the existential threat of global warming, student activists from around the world are skipping school Friday, for what organizers call a Global Climate Strike. The young activists are protesting as the U.N. prepares to hold its Climate Action Summit on Monday in New York City. The strike's figurehead is 16-year-old Swedish activist Greta Thunberg, who traveled from Sweden to New York on an emissions-free sailboat. A little over a year ago, Thunberg began her school strike for the climate by herself, outside the Swedish Parliament. Support for a school climate strike has since spread across the globe. In the past year, Thunberg has been nominated for the Nobel Peace Prize by Norwegian lawmakers. She's also met with Pope Francis and lawmakers in several countries. "We are currently on track for a world that could displace billions of people from their homes," Thunberg warned this week as she accepted Amnesty International's Ambassador of Conscience Award. She ended her acceptance speech with a call to action: "See you on the streets!" ENVIRONMENT AND ENERGY COLLABORATIVE Rising Generation Asserts Itself On Climate ChangeIn New York City, thousands of students are expected to fill the streets alongside Thunberg because, as the city's school district announced on Twitter, it is giving strikers excused absences. In Oregon, Portland Public Schools is doing the same. Strikes also are planned in rural areas where just a few dozen protesters are expected. Nicholas DuVernay, 17, organized a protest in his politically conservative small town of La Grande, Ore. "Since, probably, the beginning of my junior year in high school I've been interested in climate science and pretty passionate about environmental topics," says DuVernay, who plans to study climate science when he attends college next year. A Washington Post-Kaiser Family Foundation poll this week shows a majority of teenagers believe human-caused climate change will cause harm to them. And a quarter of the poll respondents said they have participated in a school walk-out, a rally or contacted a government official on the issue. There have been similar student events in the past year. But this time, students are asking adults to join them. At the University of Nevada, Reno, Stallar Lufrano-Jardine, 36, is setting up an event on the campus where she's an employee and student. "I'm bothered by the lack of movement to make meaningful advances to solve the climate crisis," says Lufrano-Jardine. But it's clear younger people are leading this movement. And they say most adults — especially policy-makers — are moving far too slowly. Strike organizers have a list of demands that includes "respect of indigenous land, sustainable agriculture, protecting biodiversity, environmental justice and a just transition away from fossil fuels and towards renewable energy," said 17-year-old Baltimore resident and organizer Nadia Nazar. Many of those demands are part of the Green New Deal, which was crafted by progressive Democratic lawmakers but so far hasn't gone anywhere in Congress. At a Capitol Hill press event this week, Nazar said she hopes the proposal defines her generation. "I am not a part of Generation Z. I am a part of Gen GND — the generation of the Green New Deal," she said as supporters cheered. Also on Capitol Hill this week, Thunberg and other activists testified before lawmakers. Louisiana Republican Rep. Garret Graves told them that climate change has exacerbated the loss of his state's coastline. "I agree that we need to take aggressive action. I agree that we need to ensure that we move forward in a sustainable, rational manner," Graves said. But his idea of what that means is very different than the activists' vision. For instance, Graves agrees with President Trump on the need to withdraw the U.S. from the Paris climate agreement. Graves told the student organizers the pact allows China to continue emitting more carbon dioxide while the U.S. cuts emissions. "Paris and its related pledges would undermine U.S. competitiveness," his spokesman said. Graves got immediate pushback from the young activists, including 17-year-old Jamie Margolin from Seattle, who asked how Graves will respond to questions from his children and grandchildren about whether he did enough to address climate change. "Can you really look them in the eye and say, 'No, sorry, I couldn't do anything because that country over there didn't do anything, so if they're not going to do it then I'm not.' That is shameful and that is cowardly," Margolin said. Organizers are saying this climate strike will be the largest yet. More than 2,000 scientists around the world have pledged to join. Some companies also have signed on, including Patagonia and Seventh Generation. |
AuthorArticle on Climate Change Stike written by Jeff Brady at National Public Radio, Picture taken by Susan Walsh Archives
March 2021
Categories |
 RSS Feed
RSS Feed
